By: Sarah Carter, Duong Huynh, and Phoebe Seltzer
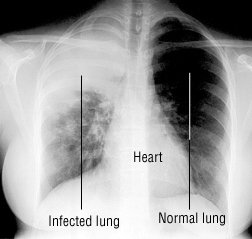 |
| Pneumonia-infected lung vs. a normal lung. Source |
Streptococcus pneumoniae is a global killer causing serious respiratory infections. Collectively, this one bacterium causes 826,000 deaths annually, mostly in young children and the elderly – and, in many ways, it’s just getting started. Improper use of broad-spectrum antibiotics has propelled S. pneumoniae into the spotlight as a very real threat for us all, with resistant strains on the rise worldwide.
Especially sly, S. pneumoniae is a master of disguise, possessing a thick protective polysaccharide layer called a capsule that comes in over 90 different varieties. Each of these 90 capsule disguises, called serotypes, possesses a unique combination of sugars, lipids, and surface proteins that grant each serotype unique abilities to evade antibiotics or a host’s immune system.
But the story has not proven to be so simple. The excellent benefits of stockpiling an unique arsenal of capsules are not without its vulnerabilities. Dr. Haley Echlin and her collaborators discovered a quirky characteristic of S. pneumoniae capsule armor - its Achilles heel lies in the capsule itself.
To understand what Echlin et al. discovered in S. pneumoniae, we must first take a step back to a molecule called hydrogen peroxide. Hydrogen peroxide is produced naturally as a byproduct of cellular metabolism, but can be dangerous to a cell at high levels. As a potent oxidant, hydrogen peroxide can wreak havoc on cells, damaging DNA and in some instances induce cellular death. Some antibiotics, such as LL-37, exploit the destructive ability of hydrogen peroxide by increasing its concentrations to a deadly level. As shown in the Figure 1, hydrogen peroxide (H2O2) is a byproduct of acetyl-p synthesis and pyruvate synthesis, both of which are part of S. pneumoniae metabolic pathway towards capsule formation.
Hypothetically, cells with lower starting levels of natural hydrogen peroxide are more likely to survive than those with higher levels when LL-37 is added. To test this hypothesis, one way scientists could decrease natural hydrogen peroxide levels in a bacteria is to mess with the cellular metabolism by introducing mutations into genes coding for critical metabolism enzymes that produce hydrogen peroxide as a byproduct. Two such enzymes, SpxB and LctO, shown in Figure 1, produce hydrogen peroxide and are also intricately involved in the metabolism of the Type IV serotype capsules.
Echlin et al. dives further into the intricate relationship between these enzymes, capsule production, LL-37 susceptibility, and virulence. Would mutating spxB and lctO result in increased resistance to LL-37, as expected? Would such mutants have capsules? How pathogenic would such mutants be? Are the metabolic pathways involving SpxB and LctO the link between LL-37 resistance and a capsule-less state?
The authors began by first investigating the levels of hydrogen peroxide in spxB and lctO mutants with the type IV (TIGR4) and type II (D39) serotype. As expected, hydrogen peroxide production decreased greatly in these mutated strains; this was especially true in the spxB/lctO double mutants, where hydrogen peroxide levels dropped as low as 5% of the wild type.
Lowering hydrogen peroxide levels could be a good thing when you are a bacteria attacked by a drug that seeks to increase hydrogen peroxide levels in order to kill you. With this in mind, Echlin et al. measured the susceptibility of these mutants to the hydrogen peroxide-increasing LL-37 antimicrobial. A expected, both type IV spxB and lctO mutants displayed heightened resistance to LL-37. Surprisingly, however, the double mutant displayed hindered growth, suggesting it was actually MORE susceptible to LL-37 even though it had the least initial amount of hydrogen peroxide. When repeated with the type II mutants, no differences in LL-37 susceptibility were observed even though they had the same amount of initial hydrogen peroxide level as the type IV mutants. This suggests that the ability to resist LL-37 was not solely due to hydrogen peroxide production as expected, but appeared to be a unique quality of the type IV single mutants.
The authors then investigated the role of spxB and lctO in capsule biosynthesis. Previous studies showed that bacteria without a capsule were more resistant to antibiotics such as LL-37. Echlin et al. proposed that a possible reason for this observation is that deletion of spxB causes downstream effects on capsule biosynthesis. To investigate this, they created different mutants to see if spxB is critical to capsule production. When spxB was mutated in a type IV serotype, it did not form a capsule. In contrast, the lctO mutant of the same serotype had an identical amount of capsule as the wild type type IV serotype. The double mutant, which had both these mutations, had the same amount of capsule as the wild type. Surprisingly, in the type II background, the capsule production was not affected in any of the mutants. It seemed something intriguing was going on with the type II bacteria...
Since differences in capsule production were observed in different backgrounds, this suggested that deletion of spxB affects capsule production differently in type II compared to type IV serotypes. To confirm this hypothesis, the authors gave the TIGR4 strain a type II capsule, and the D39 strain a type IV capsule. As expected, the TIGR4 spxB mutant with the type II capsule preserved its capsule, while the D39 spxB mutant with the type IV capsule did not. This suggests that the spxB mutation affects capsule production in a serotype specific manner, rather than a strain specific manner. Like a hole in a knight’s armor, the deletion of spxB had explosive effects on the type IV capsule. The type II, however, like an upgraded version of armor with stronger metal and more complete coverage, does not include this weakness. Give a knight, or in this case, a strain of S. pneumoniae, the new armor - and he is protected. Give him the old one with the hole - and he is once again vulnerable to the effects of spxB deletion.
| A knight is only as good as her armor: Brienne, from Game of Thrones. Source |
Next, the authors further confirmed their results with fluorescence microscopy. As shown in Figure 4A and C, the TIGR4 (type IV) wild type and the lctO mutant fluoresced. However, there was very little if any fluorescence in the spxB mutant meaning that capsule biosynthesis was negatively disrupted (Figure 4B). There was an interesting result in the double mutant where the fluorescence was not as widely distributed in this mutant as the wild type, which means that the capsule was not as widely distributed (Figure 4D). In the D39 (type II) background, all mutants had wild type levels of fluorescence (Figure 4E-H). These results show that the deletion of spxB causes a defect in capsule biosynthesis only in the TIGR4 serotype and not in the D39 serotype.
The research team then examined acetyl-CoA, an influential aspect of S. pneumoniae glucose metabolism and capsule production. By deleting spxB in S. pneumoniae, acetyl-CoA levels decreased and capsule production was negatively disrupted. BUT this result was specific to strains of the type IV serotype that have acetylated-sugars in their capsules, including TIGR4, because acetyl-CoA is a necessary cofactor for capsule production in these particular strains. S. pneumoniae mutants that do not have acetylated sugars in their capsules, such as D39, are not affected by the change in acetyl-CoA levels, and thus their capsule synthesis are not affected by the spxB mutation. Targeting acetyl-CoA related metabolism may not be an efficient move if the goal is to fight global S. pneumoniae virulence, but could be useful in fighting specific strains with a specific capsule type rather than the entire army of diverse S. pneumoniae.
 |
| Source |
In order to test the virulence of these type IV mutant strains, lab mice were injected with wild type and the TGIR4 spxB mutant. Good news, the TIGR4 spxB mutants (those that lacked the spxB gene) were less virulent than the wild type bacteria. The unlucky mice who were given the wild type strain were inundated by a larger population of colonies that had formed over time. The TIGR4 spxB mutants also allowed the mice to live longer than the wild-type infected mice. Even better, TIGR4 spxB mutants were not deadly, and this may be a result of reducing S. pneumoniae capsule synthesis. Thus, even though the TIGR4 spxB are more resistant to the antimicrobial LL-37, this strain is also less virulent. So even if drug companies will not make a drug to disrupt S. pneuomiae’s spxB gene, we now know that if you are ever infected with pneumoniae, pray that it’s the TIGR4 spxB mutant. Diversity is S. pneumoniae’s greatest weapon, and if you’re lucky, its weakness.
Let’s take a moment to praise the beauty and intelligence of S. pneumoniae. It’s diverse and inclusive to strains of all capsule type and metabolic levels. It’s also deadly! While hydrogen peroxide levels don’t seem to be the bacterial Rosetta Stone to understanding S. pneumoniae’s antimicrobial defense mechanisms, its capsule plays an intricate role in its pathogenic abilities. For humans, we might as well give up on the search for a cure to the more murderous strains, but then again, maybe there will be some other weakness shared by all serotypes in the S. pneumoniae army that are targetable. Perhaps if we target an earlier stage metabolic product, something that is necessary for metabolism and capsule synthesis in all serotypes, then we can efficiently kill them all. Meanwhile hundreds of thousands of people will continue to suffer from S. pneumoniae, but the search is on!
| Benedict Cumberbatch as Sherlock Holmes in the BBC’s Sherlock, searching for answers. Source |
| A healthy pair of lungs. Source |
More Information: Echlin et al. (2016) Pyruvate oxidase as a critical link between metabolism and capsule biosynthesis in Streptococcus pneumoniae. PLoS Pathogens 12: e1005951. https://doi.org/10.1371/journal.ppat.1005951
~~
 |
| Meet the authors: Phoebe Seltzer (L), Duong Huynh (M), and Sarah Carter (R) |
Phoebe is a junior at Mount Holyoke College major in Biochemistry from Sudbury, MA. She enjoys running, and is currently training for her first ultramarathon. Duong Huynh is a senior majoring in Biology and minoring in English at MHC. She enjoys listening to music, podcasts, surfing the web, and writing fictional stories. Sarah is a senior at Mount Holyoke College majoring in Biochemistry and minoring in Music. She has a broad range of interests, from biochemistry to organoids to playing the upright and electric bass. Upon graduation from Mount Holyoke, Sarah will be attending Utrecht University in the Netherlands to pursue a Master’s degree in Cancer, Stem Cells, and Developmental Biology.


No comments:
Post a Comment